 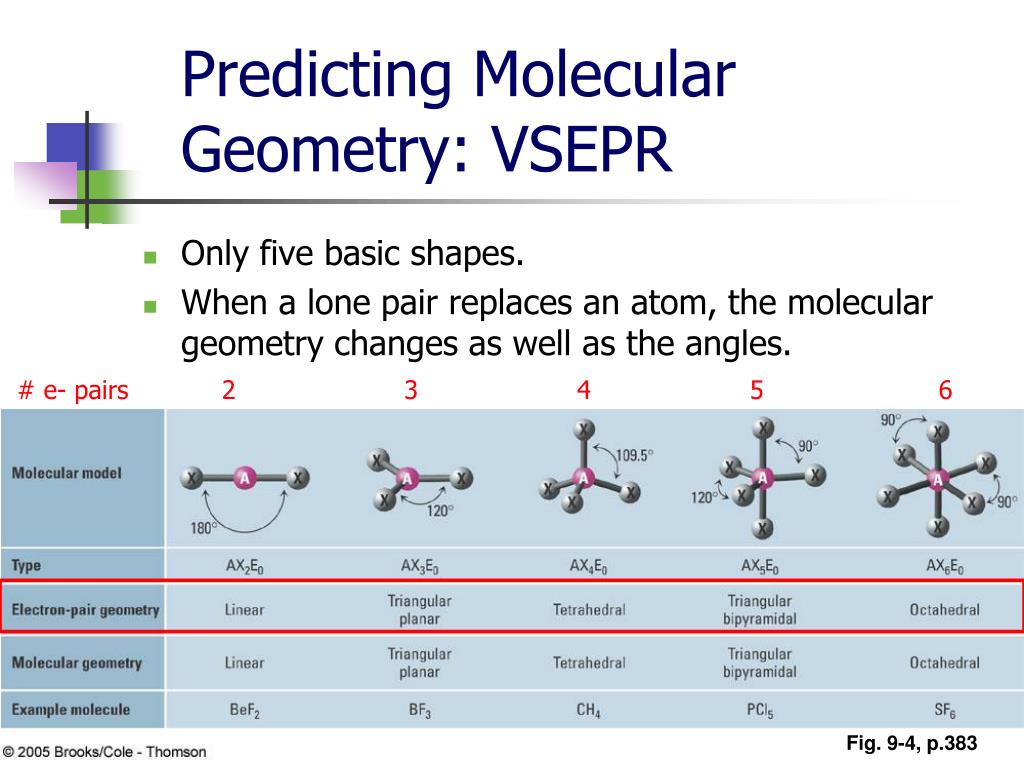 
Typically the bond distance to the axial ligands is longer than to the equatorial ligands. The equatorial pair of ligands is situated in a plane orthogonal to the axis of the axial pair. The axial pair lie along a common bond axis so that are related by a bond angle of 180°. Repulsion by bonding pairs at 120° is much smaller and less important.Ĭompounds with disphenoidal (see-saw) geometry have two types of ligands: axial and equatorial. An equatorial lone pair is repelled by only two bonding pairs at 90°, whereas a hypothetical axial lone pair would be repelled by three bonding pairs at 90° which would make it stable. This is true because the lone pair occupies more space near the central atom (A) than does a bonding pair of electrons. An atom bonded to 5 other atoms (and no lone pairs) forms a trigonal bipyramid with two axial and three equatorial positions, but in the seesaw geometry one of the atoms is replaced by a lone pair of electrons, which is always in an equatorial position. The seesaw geometry occurs when a molecule has a steric number of 5, with the central atom being bonded to 4 other atoms and 1 lone pair (AX 4E 1 in AXE notation). 
Most commonly, four bonds to a central atom result in tetrahedral or, less commonly, square planar geometry. The name "seesaw" comes from the observation that it looks like a playground seesaw. Ideal ax-ax 180°, eq-eq 120°, ax-eq 90° SF 4 ax-ax 173.1°, eq-eq 101.6°ĭisphenoidal or seesaw (also known as sawhorse) is a type of molecular geometry where there are four bonds to a central atom with overall C 2v molecular symmetry. A computational chemistry study showed a distorted planar Y-shaped geometry with the smallest F–Xe–F bond angle equal to 69°, rather than 90° as in a T-shaped geometry.Structural molecular geometry Seesaw molecular geometry Although this anion has been detected in the gas phase, attempts at synthesis in solution and experimental structure determination were unsuccessful. ģ, has been investigated as a possible first example of an AX 3E 3 molecule, which might be expected by VSEPR reasoning to have six electron pairs in an octahedral arrangement with both the three lone pairs and the three ligands in a mer or T-shaped orientations. The three atoms bond at 90° angles on one side of the central atom, producing the T shape. In an AX 3E 2 molecule, the two lone pairs occupy two equatorial positions, and the three ligand atoms occupy the two axial positions as well as one equatorial position. The T-shaped geometry is related to the trigonal bipyramidal molecular geometry for AX 5 molecules with three equatorial and two axial ligands. Īccording to VSEPR theory, T-shaped geometry results when three ligands and two lone pairs of electrons are bonded to the central atom, written in AXE notation as AX 3E 2. Examples of T-shaped molecules are the halogen trifluorides, such as ClF 3. Ordinarily, three-coordinated compounds adopt trigonal planar or pyramidal geometries. In chemistry, T-shaped molecular geometry describes the structures of some molecules where a central atom has three ligands. 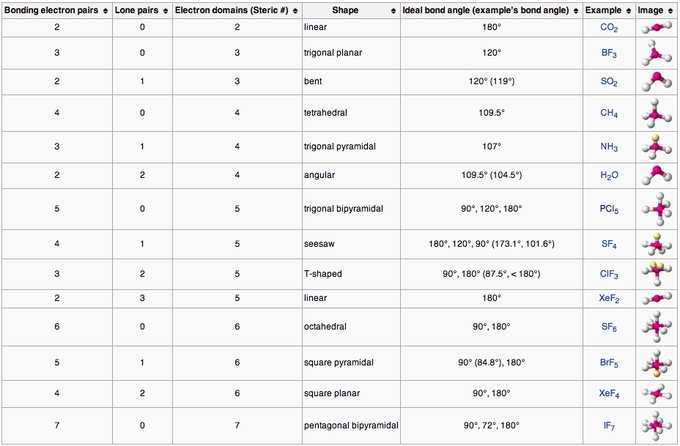
Structure of chlorine trifluoride, an example of a compound with T-shaped coordination geometry. Type of molecular geometry T-shaped molecular geometry 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
